Carbon and its allotropes are resistant to melting at atmospheric pressure and remain solid at considerably high temperature. All allotropes of carbon are solid under standard conditions. Graphite is the most thermodynamically stable allotrope of carbon at standard temperature and pressure. Elemental carbon is insoluble in water, organic solvents and acids and bases. Carbon and its allotropes have the highest thermal conductivities as compared to all elements. The electrical conductivity of graphite is very good as compared to diamond, which is an outstanding electrical insulator. Graphite is black, opaque and extremely soft, while crystal is highly transparent and is the hardest natural element. The different allotropic forms of carbon have different physical properties. Natural reserves of diamond are present in Canada, Congo, Russia and South Africa. Carbon is also present in stars and sun and in the atmosphere of many planets. Carbon is major hydrocarbons present in fossil fuels including coal, natural gas, and crude oil. The common allotropes of carbon include diamond, graphite, amorphous carbon (charcoal and carbon black) and nanoforms of carbon including graphene and fullerenes. Carbon exists in various forms, termed as allotropes (variation in bonding of carbon atoms due to its valency). Atomic form of carbon is very short lived and readily stabilizes in multi-atomic configurations. In humans, carbon makes about 18.5% of body mass and is the second most abundant element in the body. Biologically, carbon holds a significant position and is part of all living systems. Carbon Periodic Table ClassificationĬarbon is characterized as the 4 th most abundant element in the universe and the 15 th most abundant element in the Earth’s crust. Vandermonde, Claude Louis Berthollet, and Gaspard Monge confirmed that graphite was a form of carbon in the same way as diamond (discovered earlier in 1772). Carbon was discovered as a novel element by 1722 by Antoine Ferchault de Réaumur, who proposed that this novel element can be used to transform iron into steel. The name carbon has been derived from the word carbo (Latin for coal and charcoal). 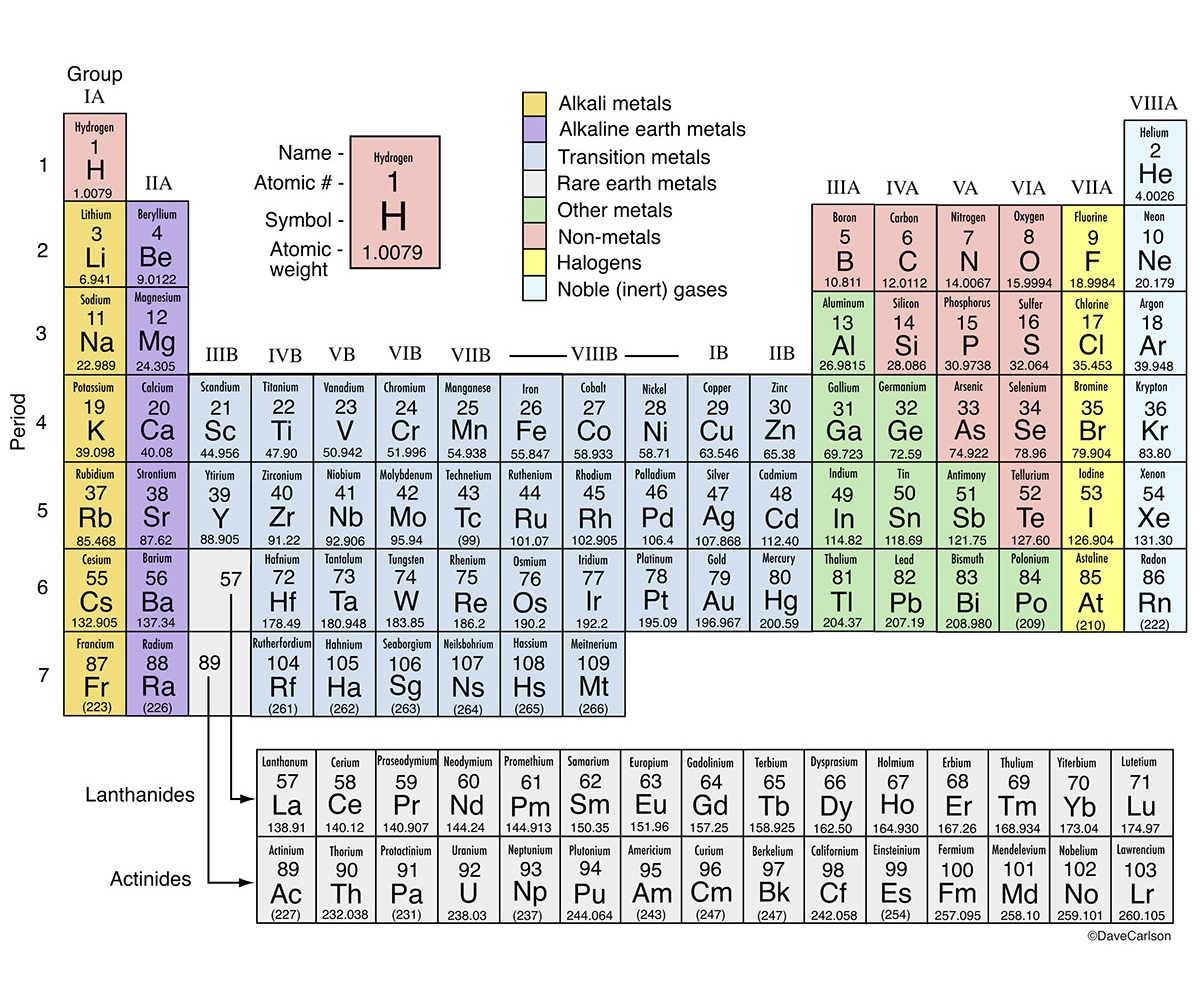
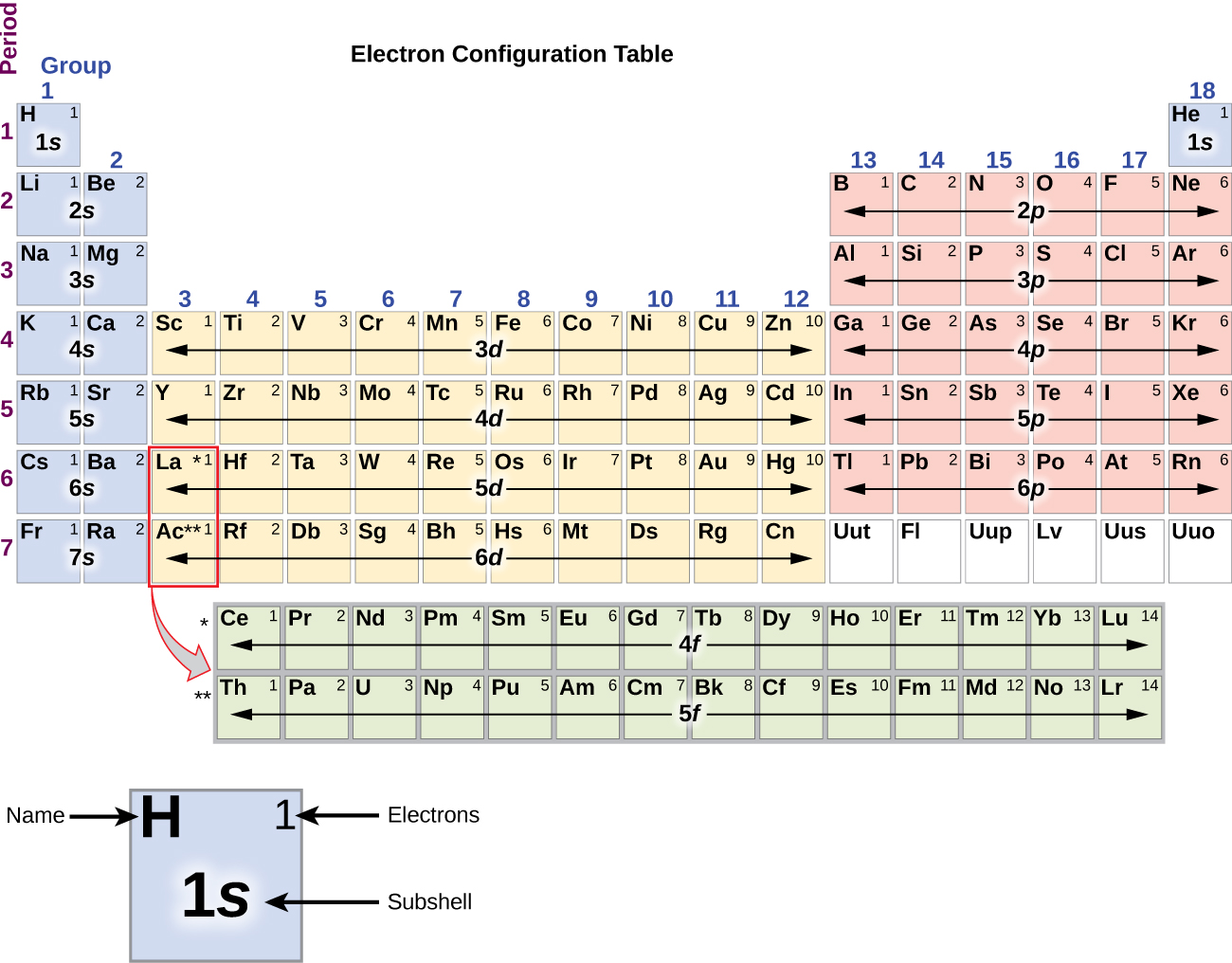
Diamonds were discovered in China in around 2500BCE. The discovery of carbon dates to prehistoric times and was used by ancient human civilizations as in the form of charcoal and coal. It belongs to group 14 of the periodic table. It is nonmetallic and tetravalent-making four electrons available to form covalent chemical bonds. Magnesia, a district of Eastern Thessaly in GreeceĪlumina, from Latin alumen (gen.Carbon is a chemical element with symbol C and atomic number 6. Symbol Na is derived from Neo-Latin natrium, coined from German Natron, ' natron'.Greek elements hydro- and -gen, ' water-forming'īeryl, a mineral (ultimately from the name of Belur in southern India) īorax, a mineral (from Arabic bawraq, Middle Persian * bōrag) For more detailed information about the origins of element names, see List of chemical element name etymologies. Like the periodic table, the list below organizes the elements by the number of protons in their atoms it can also be organized by other properties, such as atomic weight, density, and electronegativity. It is a tabular arrangement of the elements by their chemical properties that usually uses abbreviated chemical symbols in place of full element names, but the linear list format presented here is also useful. The definitive visualisation of all 118 elements is the periodic table of the elements, whose history along the principles of the periodic law was one of the founding developments of modern chemistry. A chemical element, often simply called an element, is a type of atom which has the same number of protons in its atomic nucleus (i.e., the same atomic number, or Z). This is a list of the 118 chemical elements that have been identified as of 2023. List of the 118 identified chemical elements
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
